Bacteriostic Water 30ml (0.9% Benzyl Alcohol)
$28.00
| Quantity | Discount | Price |
|---|---|---|
| 5 - 8 | 5% | $26.60 |
| 9+ | 10% | $25.20 |
Free shipping on orders over $200!
- Satisfaction Guaranteed
- No Hassle Refunds
- Secure Payments
Bacteriostatic Water (0.9% Benzyl Alcohol)
Bacteriostatic Water for Injection (BWFI) is a sterile, non-pyrogenic preparation of water containing 0.9% benzyl alcohol, functioning as a bacteriostatic preservative. It is widely used in research settings for dilution, reconstitution, and suspension of peptides, laboratory compounds, and biological materials intended strictly for experimental applications. The presence of benzyl alcohol inhibits bacterial growth in multi-dose containers, making it suitable for repeated withdrawals under aseptic conditions.
Specifications
Synonyms: BWFI, Bacteriostatic Water USP, Bacteriostatic Water for Injection
Components: Sterile water + 0.9% benzyl alcohol (preservative)
pH Range: Typically 4.5–7.0 (USP standard)
Osmolality: Hypotonic solution
Container Type: Multi-dose vial (intended for repeated puncture under sterile technique)
Class: Sterile aqueous diluent / preservative-containing solvent for laboratory use
Mechanism of Action and Antimicrobial Activity
Bacteriostatic Water contains 0.9% benzyl alcohol, which provides bacteriostatic activity by disrupting bacterial cell membranes and protein processes. Published work describes benzyl alcohol as:
A membrane-penetrating aromatic alcohol capable of destabilizing lipid bilayers
An inhibitor of bacterial metabolic enzymes
A preservative effective against a wide spectrum of microorganisms in aqueous formulations
Studies show that benzyl alcohol’s antimicrobial effectiveness in multi-dose vials prevents microbial proliferation after repeated needle entries—provided aseptic technique is maintained.
Use in Laboratory Reconstitution and Dilution
In research environments, bacteriostatic water is commonly employed to:
Reconstitute lyophilized peptides for in vitro assays
Prepare solutions for ex vivo tissue studies
Maintain sterility during multiple withdrawals from the same container
Extend viability of aqueous experimental materials by preventing microbial contamination
Bacteriostatic preservatives significantly reduce microbial contamination risk in multi-use containers, according to microbiological stability research evaluating benzyl alcohol–containing diluents.
Stability, Storage, and Handling
Stability considerations:
Benzyl alcohol provides antimicrobial protection, not sterilization—solutions remain sterile only if handled aseptically.
Exposure to air, repeated puncture without sterile technique, or improper storage can compromise sterility.
BWFI should be discarded if particulate matter, discoloration, or visible contamination is observed.
Storage:
Maintain at controlled room temperature (20–25°C per USP guidance).
Protect from extreme heat or freezing.
Compatibility:
Compatible with many experimental peptides and biological compounds.
Should not be used with substances known to be benzyl alcohol–sensitive in cell assays.
Safety and Toxicology Notes (Research Context Only)
Toxicological literature describes benzyl alcohol as:
A metabolic substrate oxidized to benzoic acid
Generally low-toxicity in controlled laboratory concentrations
Potentially cytotoxic at high concentrations or in sensitive cell lines
Neonatal toxicity associated with intravenous benzyl alcohol exposure has been well documented in clinical contexts, reinforcing the need for strict laboratory-only use and prohibition of any human or clinical application.
Other Experimental Applications
Bacteriostatic water has been used in laboratory studies evaluating:
Preservative efficacy testing in multi-dose pharmaceutical systems
Microbial challenge assays assessing benzyl alcohol’s inhibitory spectrum
Drug stability and solubility studies, where sterile aqueous vehicles are required
Preparation of calibration standards and buffered solutions for analytical methods
Multiple publications identify benzyl alcohol as a model compound for studying preservative–membrane interactions and physicochemical stability in aqueous formulations.
Research Use Only – Important Notice
This Bacteriostatic Water product is supplied exclusively for laboratory research purposes.
Not for human or veterinary use
Not for diagnostic, therapeutic, or clinical applications
Intended only for in vitro and controlled experimental use
Must be handled with sterile technique according to institutional laboratory standards
All descriptions above summarize findings from microbiological and pharmaceutical-science studies and must not be interpreted as medical guidance or instructions for clinical use.
References
U.S. Pharmacopeia (USP). Bacteriostatic Water for Injection – Specifications and Benzyl Alcohol Preservative Requirements. Available at:
https://www.uspnf.comMeyer BK et al. Benzyl alcohol as a preservative: antimicrobial activity and physicochemical properties. J Pharm Sci. Available at:
https://pubmed.ncbi.nlm.nih.govFDA Labeling Database – Bacteriostatic Water for Injection (benzyl alcohol 0.9%): stability, antimicrobial properties, and multi-dose vial usage. Available at:
https://www.accessdata.fda.gov/drugsatfda_docsGagnon M et al. Preservative efficacy of benzyl alcohol in aqueous multi-dose systems. PDA J Pharm Sci Technol. Available at:
https://pubmed.ncbi.nlm.nih.gov
| ML | 3ml, 10ml, 30ml |
|---|









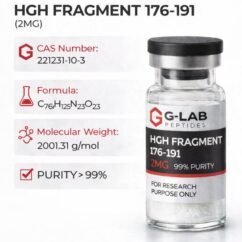


 DSIP (5mg)
DSIP (5mg) 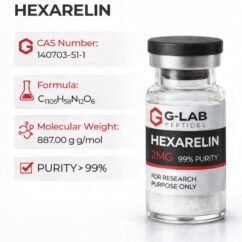 Hexarelin (2mg)
Hexarelin (2mg)