Melanotan 2 (10mg)
$48.90
| Quantity | Discount | Price |
|---|---|---|
| 5 - 8 | 5% | $46.46 |
| 9+ | 10% | $44.01 |
Free shipping on orders over $200!
- Satisfaction Guaranteed
- No Hassle Refunds
- Secure Payments
Melanotan II (10 mg)
Melanotan II (MT-II) is a synthetic cyclic heptapeptide analog of the endogenous peptide hormone α-melanocyte-stimulating hormone (α-MSH). It was designed as a super-agonist at melanocortin receptors, particularly MC1R and MC4R, with enhanced in vivo stability and prolonged duration of action compared with native α-MSH. UTUPub+3DNB Portal+3ResearchGate+3
By activating melanocortin receptors, Melanotan II has been shown in preclinical and early clinical studies to:
Induce melanogenesis and increase cutaneous pigmentation (tanning) via MC1R on melanocytes
Modulate sexual function and arousal, including pro-erectile effects, via central MC3R/MC4R pathways
Influence energy balance, appetite and autonomic function in metabolic and cardiovascular models
Melanotan II is not an approved medicine; unregulated products containing MT-II (injectables and nasal sprays) marketed for tanning and sexual enhancement have raised significant safety and regulatory concerns worldwide. New York Post+6HPRA+6Therapeutic Goods Administration (TGA)+6
Specifications
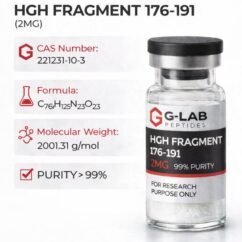
Synonyms: Melanotan II, MT-II, MT2, MT-2, [Ac-Nle-c(Asp-His-D-Phe-Arg-Trp-Lys)-NH₂], α-MSH analog
Sequence:
Ac-Nle-cyclo[Asp-His-D-Phe-Arg-Trp-Lys]-NH₂ (lactam bridge between Asp and Lys) DNB Portal+2MedChemExpress+2
Molecular formula: C₅₀H₆₉N₁₅O₉ GenScript
Molecular weight: 1024.18 g/mol GenScript+1
Class: Synthetic melanocortin receptor agonist (α-MSH analog; cyclic heptapeptide super-agonist) OUP Academic+2mediaTUM+2
Presentation (research grade):
Lyophilized powder, 10 mg per vial
Reconstitution solvent and working concentrations depend on experimental design and must follow lab-specific protocols.
Mechanism of Action and Melanocortin Signaling
Melanotan II acts as a non-selective melanocortin receptor agonist, with high affinity for MC1R, MC3R, MC4R and MC5R. UTUPub+2ScienceDirect+2
Key mechanistic features include:
MC1R activation – melanogenesis
MT-II mimics α-MSH on melanocortin 1 receptor (MC1R) expressed on melanocytes, increasing melanin synthesis and dispersion, leading to darker skin pigmentation in response to UV exposure and, to a lesser extent, even at baseline. DermNet®+3PubMed+3Actas Dermo-Sifiliográficas+3
MC4R / MC3R activation – central nervous system effects
Reviews and clinical work indicate that MC4R and MC3R in the hypothalamus and other CNS regions mediate sexual arousal, erectile responses, appetite and autonomic tone. OUP Academic+3ScienceDirect+3European Urology+3
MT-II crosses the blood–brain barrier and acts centrally to trigger penile erection and increased sexual desire independent of (but augmented by) sexual stimulation. PubMed+4mediaTUM+4PubMed+4
Super-agonist properties
Structural cyclization (Asp–Lys lactam) and N-terminal acetylation confer greater potency and stability than α-MSH and many linear analogs, with nanomolar affinities at MC1R and MC4R. DNB Portal+2OUP Academic+2
Overall, Melanotan II provides a robust tool to activate the melanocortin system in vivo and in vitro, making it useful for research on pigmentation, sexual function, energy balance and cardiovascular regulation.
Skin Pigmentation and Photobiology Research
Melanotan II has been widely used to study melanogenesis and photoprotection:
Early experimental and small clinical studies showed that low subcutaneous doses of MT-II can increase skin pigmentation over days to weeks, often with less UV exposure than needed for equivalent tanning in untreated subjects. DermNet®+3DNB Portal+3ResearchGate+3
The peptide promotes eumelanin production, which is associated with darker, more photoprotective pigment, and can be used as a research tool to explore how increased baseline pigmentation affects UV-induced DNA damage and photocarcinogenesis in experimental systems. DNB Portal+2Actas Dermo-Sifiliográficas+2
Observational work on unregulated online MT-II products used by consumers has documented variable dose regimens, injection or nasal administration, and frequent side effects such as nausea, flushing and changes in existing naevi, with concerns about potential melanoma risk, particularly in the setting of high UV/sunbed exposure. New York Post+7JAAD+7PubMed+7
Sexual Function and Neuroendocrine Research
Melanotan II has played a central role in uncovering the link between melanocortin signaling and sexual function:
Erectile dysfunction (ED) trials
In a double-blind, placebo-controlled crossover study, subcutaneous MT-II initiated penile erection in 17 of 20 men with psychogenic or organic ED without the need for explicit sexual stimulation, with mean episodes of high-rigidity erections recorded by RigiScan. Gold Journal+3PubMed+3Nature+3
A dose of 0.025 mg/kg produced robust erectile responses but was frequently associated with nausea and yawning, with ~13% of subjects reporting severe nausea at that dose. PubMed+2PubMed+2
Sexual desire and arousal
Hadley and colleagues reported that MT-II enhanced erectile activity in men and sexual desire and genital arousal in women, suggesting a broader role for central melanocortin pathways in human sexual behavior. PubMed+2PMC+2
Mechanistic insights
Reviews of melanocortin receptor agonists highlight MC4R as a key mediator of pro-erectile and pro-sexual effects, though the exact neuronal circuits remain incompletely defined. ScienceDirect+2European Urology+2
These studies make Melanotan II a highly informative tool compound for exploring central melanocortin regulation of sexual function, while underscoring that MT-II itself has not been developed as an approved ED treatment (unlike its derivative bremelanotide).
Energy Balance, Metabolism and Cardiovascular Research
Activation of the melanocortin system has profound effects on energy homeostasis and autonomic function:
Appetite and body weight
Melanotan II and related MC agonists reduce food intake and body weight in rodent models by activating hypothalamic MC3R/MC4R, providing insight into melanocortin control of obesity and cachexia. UTUPub+3OUP Academic+3ScienceDirect+3
Atherosclerosis and vascular function
In ApoE⁻/⁻ mice, low-dose MT-II treatment improved lipoprotein profiles, reduced vascular inflammation and atherosclerotic lesion burden, supporting a role for melanocortin signaling in vascular homeostasis. AHA Journals+1
Autonomic / cardiovascular effects
Melanocortins influence blood pressure and sympathetic tone; MT-II has been used to dissect these actions in preclinical models, though its pressoric and tachycardic effects raise safety concerns outside controlled research settings. ScienceDirect+2AHA Journals+2
In sum, Melanotan II is a valuable experimental agonist to interrogate central melanocortin pathways linking energy balance, cardiovascular regulation and behavior.
Safety, Adverse Effects and Regulatory Warnings
Acute adverse effects (from trials and case reports)
Common dose-limiting effects:
Nausea, facial flushing, yawning, stretching, and transient fatigue were the most frequent adverse effects in ED trials and pigmentation studies. JAAD+4PubMed+4PubMed+4
Serious and rare events (case reports):
Systemic toxicity with sympathomimetic excess and rhabdomyolysis has been described following self-administered MT-II obtained online. PubMed
Priapism and severe penile pain have been reported after non-medical Melanotan II use for tanning and sexual enhancement. ScienceDirect
Pigmentary changes:
Clinicians report rapid development or darkening of melanocytic naevi and lentigines in MT-II users, prompting concern about melanoma risk, particularly under heavy UV exposure. Long-term carcinogenic risk remains uncertain, but regulatory agencies recommend avoiding use. New York Post+5Actas Dermo-Sifiliográficas+5DermNet®+5
Regulatory and public-health statements
National agencies (e.g., HPRA, TGA and others) firmly state that Melanotan II is an unapproved, unsafe substance, often sold illegally online as tanning injections or nasal sprays. The Courier-Mail+5HPRA+5Therapeutic Goods Administration (TGA)+5
Health authorities warn of variable product quality, contamination, unknown dosing and unstudied long-term risks, and advise consumers to avoid MT-II in favor of regulated cosmetic self-tanning products that do not alter systemic physiology. The Courier-Mail+4Therapeutic Goods Administration (TGA)+4The Guardian+4
These findings strongly support restricting Melanotan II to controlled laboratory and preclinical research, not human self-administration.
Other Experimental Applications
Beyond pigmentation and sexual function, Melanotan II is used in diverse experimental contexts:
Receptor pharmacology and structure–activity studies:
MT-II is a reference agonist in binding and signaling assays for MC1R, MC3R, MC4R and MC5R, aiding development of more selective melanocortin drugs (e.g., afamelanotide, bremelanotide). UTUPub+3OUP Academic+3Science.gov+3
Imaging and tumor targeting:
Radiolabeled MT-II analogs are used for melanoma targeting and imaging, taking advantage of MC1R overexpression in melanoma cells. Science.gov+1
Neurobehavioral research:
MT-II has been applied in models of stress, anxiety, reward and feeding, helping to map melanocortin contributions to central emotional and behavioral circuits. AHA Journals+3OUP Academic+3ScienceDirect+3
These applications highlight Melanotan II as a versatile probe for melanocortin biology across dermatology, endocrinology, neuroscience and cardiometabolic research.
Research Use Only – Important Notice
This Melanotan II (10 mg) product is supplied exclusively for laboratory research purposes.
Not for human or veterinary use
Not for diagnostic, therapeutic, tanning, performance-enhancing or cosmetic applications
Intended only for in vitro experiments and/or appropriately controlled animal studies conducted by qualified professionals
All descriptions above summarize findings from preclinical and limited early human studies and regulatory / public-health communications.
They must not be interpreted as medical claims, dose recommendations, or any form of guidance for self-administration or clinical use.
References
Ryakhovsky VV. et al. The first preparative solution phase synthesis of melanotan II. Beilstein J Org Chem. 2008. DNB Portal+1
GenScript Catalog. Melanotan II – peptide information (sequence, formula, MW). GenScript
Wardlaw SL. Obesity as a neuroendocrine disease: lessons from melanocortin receptor-deficient mice. J Clin Endocrinol Metab. 2001. OUP Academic
Rinne P. New aspects on the melanocortins and their cardiovascular actions. PhD thesis, Univ. of Turku. 2012. UTUPub+1
Martin WJ. Melanocortin receptors and erectile function. Eur Urol. 2004. ScienceDirect+1
Wessells H. et al. Synthetic melanotropic peptide initiates erections in men with psychogenic erectile dysfunction. J Urol. 1998; and follow-up melanocortin agonist trials. Gold Journal+5PubMed+5ScienceDirect+5
Hadley ME. et al. Discovery that a melanocortin regulates sexual functions in humans. Ann N Y Acad Sci. 2005. PubMed
Gilhooley E. et al. Melanotan II user experience: A qualitative study of online discussion forums. Clin Exp Dermatol. 2021. PubMed+1
Nelson ME. et al. Melanotan II injection resulting in systemic toxicity and rhabdomyolysis. Clin Toxicol (Phila). 2012. PubMed
Mallory CW. et al. Melanotan tanning injection: A rare cause of priapism. Urol Case Rep. 2021. ScienceDirect
DermNet NZ. Melanotan II – unlicensed synthetic α-MSH analog. DermNet®
UNSW / Sebaratnam D. What is melanotan-II – the drug the TGA urges consumers to avoid? 2023. UNSW Sites
HPRA. Reminder of serious health risks with Melanotan 2 self-tan products. 2023. HPRA
TGA. Don’t risk using tanning products containing melanotan. 2025. Therapeutic Goods Administration (TGA)
Trading Standards / Melanoma Focus & media reports on melanotan nasal sprays and drops. The Courier-Mail+3The Guardian+3










 Hyaluronic Acid 2% + GHK-Cu (Serum)
Hyaluronic Acid 2% + GHK-Cu (Serum)