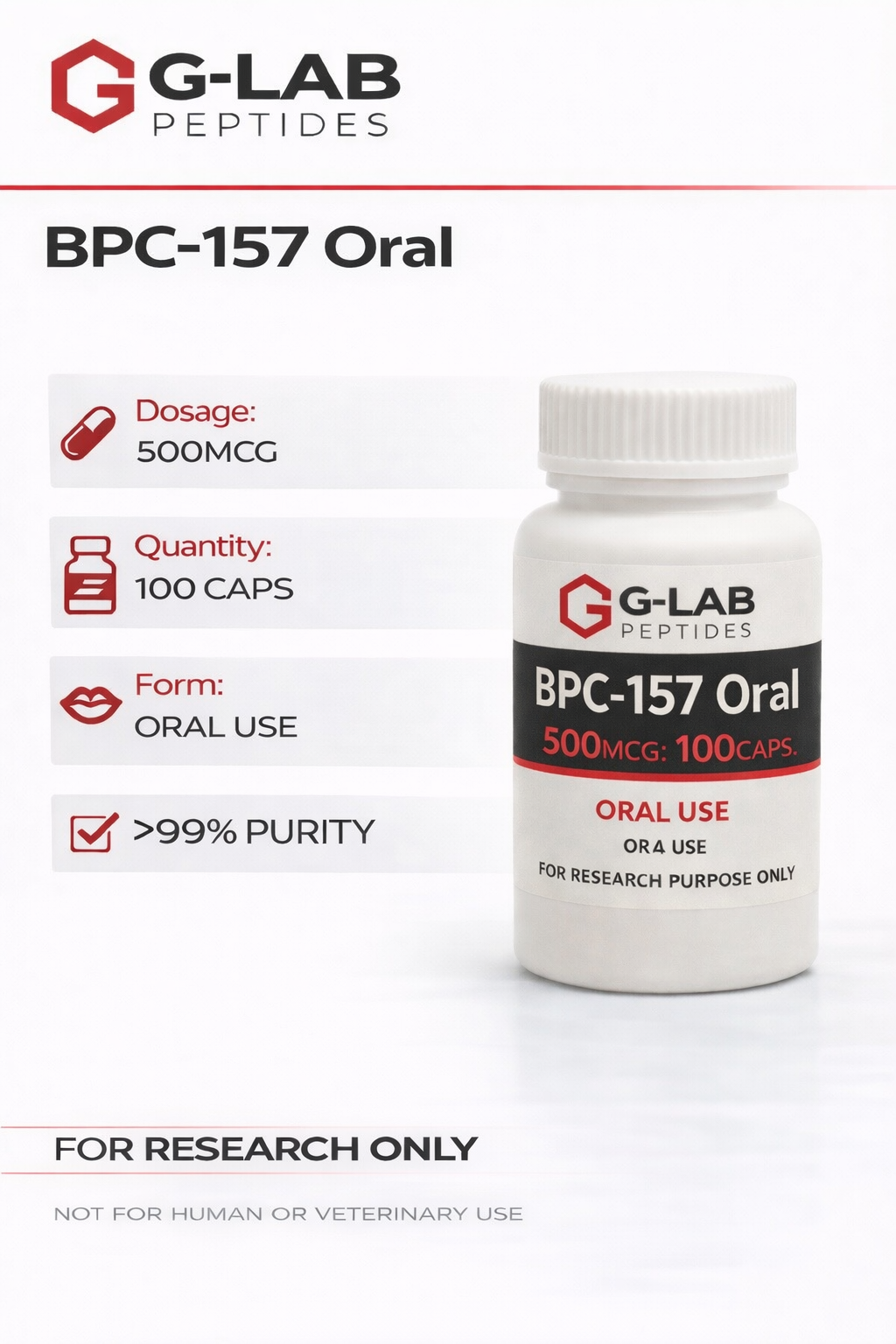
BPC-157 Oral (500mcg)
$169.90
| Quantity | Discount | Price |
|---|---|---|
| 5 - 8 | 5% | $161.41 |
| 9+ | 10% | $152.91 |
Free shipping on orders over $200!
- Satisfaction Guaranteed
- No Hassle Refunds
- Secure Payments
BPC-157 Oral – 500 mcg
BPC-157, also known as stable gastric pentadecapeptide, is a synthetic 15–amino acid fragment (sequence GEPPPGKPADDAGLV) originally isolated from a larger cytoprotective protein found in gastric juice. It has been extensively investigated in preclinical models as a potential mediator of so-called “Robert’s cytoprotection,” with proposed roles in maintaining gastrointestinal mucosal integrity, modulating the brain–gut axis, and supporting healing processes in muscle, tendon, nerve, liver, and other tissues.[1–4]
Across rodent models, BPC-157 has been reported to exert protective activity when administered via various routes, including oral, intraperitoneal, and in drinking water, and to remain stable in the gastric environment.[1,3] It has been studied as a possible experimental tool in conditions involving inflammation, ischemia–reperfusion, neurotoxicity, vascular dysfunction, and impaired tissue repair.[2,4–6]
Specifications
Synonyms:
BPC-157; Stable Gastric Pentadecapeptide; Body Protection Compound-157; PL-10; PLD-116; PL 14736
Sequence:
GEPPPGKPADDAGLV
Class:
Synthetic pentadecapeptide / cytoprotective peptide (research use only)
Molecular Weight:
≈ 1419 g/mol[1]
Molecular Formula:
C₆₂H₉₈N₁₆O₂₂
BPC-157 Research
BPC-157 and the Gastrointestinal Tract
Early work in rat gastric ulcer models demonstrated that BPC-157 may attenuate lesion formation induced by ethanol, acid, or nonsteroidal anti-inflammatory drugs (NSAIDs), while promoting faster healing of pre-existing ulcers and preserving mucosal architecture and vascular integrity.[1] In these studies, orally or intragastrically applied BPC-157 appeared to reduce hemorrhage, edema, and necrosis, with a concomitant improvement in mucosal blood flow and granulation tissue organization.[1,5]
Subsequent review articles have proposed that BPC-157 may act as a novel effector of gastric cytoprotection, involved in the maintenance of gastrointestinal mucosa under severe noxious conditions such as ischemia–reperfusion, colitis, or fistula formation.[2,3] In experimental colitis and intestinal ischemia models, BPC-157 administration has been associated with reduced macroscopic and microscopic damage, normalization of motility, and improved survival in heavily challenged animals.[4,6,7]
Mechanistically, BPC-157 has been linked to modulation of nitric oxide (NO) pathways, early growth response gene-1 (Egr-1) signaling, NAB2, focal adhesion kinase (FAK)–paxillin complexes, and JAK2/STAT pathways, all of which may contribute to cytoprotection, angiogenesis, and tissue repair in the gut.[2,8]
BPC-157 and Musculoskeletal / Tendon Healing
In musculoskeletal research, BPC-157 has been studied for its potential to accelerate tendon and muscle repair. In a classic rat model of Achilles tendon transection, systemic or local administration of BPC-157 was reported to accelerate the healing of transected tendons, improve biomechanical properties, and stimulate tenocyte outgrowth in vitro.[9]
In a related model where the Achilles tendon was sharply detached from the calcaneal bone, BPC-157 appeared to promote early functional recovery, support tendon-to-bone reattachment, and counteract the detrimental effects of high-dose corticosteroid treatment on tendon healing.[10,11] These findings suggest that BPC-157 may influence fibroblast activity, collagen remodeling, and neovascularization within the tendon–bone interface.
Other work has indicated that BPC-157 may improve muscle healing after crush injury, with enhanced myofiber regeneration, reduced edema and necrosis, and improved functional scores in treated animals.[3,11] Collectively, these data support the use of BPC-157 as a research tool to explore pathways involved in tendon regeneration, enthesis (tendon–bone junction) biology, and skeletal muscle repair.
BPC-157 and the Central Nervous System
The central nervous system (CNS) has been another major focus of BPC-157 research. In a mouse model of traumatic brain injury (TBI) induced by a falling weight, various BPC-157 regimens produced a marked attenuation of brain damage, improved early neurological outcomes, and reduced delayed mortality over the first 24 hours post-injury.[12] Histological analysis in these models indicated less edema, hemorrhage, and necrosis in BPC-157-treated animals.[12]
A more recent review dedicated to BPC-157 and the CNS summarized data from multiple models, including TBI, spinal cord trauma, stroke-like insults, seizures, and encephalopathies induced by toxins, drugs, or metabolic insults.[13] Across these diverse paradigms, BPC-157 treatment was associated with reduced lesion size, improved behavioral outcomes, and preservation of neuronal structures. The authors suggested that BPC-157 may interface with dopaminergic, serotonergic, GABAergic, and glutamatergic systems, as well as NO signaling, thereby modulating neuroinflammation, edema, and microvascular function.[2,13]
Additional studies in hepatotoxic and neurotoxic models, such as high-dose paracetamol exposure, showed that BPC-157 may counteract generalized convulsions, brain damage, and severe liver injury, further highlighting its potential relevance for research on the brain–liver axis and systemic toxicity.[14]
BPC-157, Organ Protection, and Systemic Injury
Beyond the GI tract and CNS, BPC-157 has been investigated in a range of organ injury and systemic stress models. In rats treated with the COX-2 inhibitor celecoxib, a regimen known to provoke gastrointestinal, hepatic, and brain lesions under certain conditions, BPC-157 administration appeared to markedly attenuate macroscopic and microscopic tissue damage in all three organ systems.[15] The same study suggested that BPC-157 may interact with NO pathways, since co-administration of a nitric oxide synthase inhibitor (L-NAME) aggravated lesions, whereas L-arginine or BPC-157 counteracted these effects.[15]
In ischemia–reperfusion models affecting the gut and other organs, BPC-157 treatment has been reported to improve microvascular patency, reduce thrombosis, preserve endothelial integrity, and limit the extent of necrosis and edema.[4,6,7,16] These findings have led authors to propose that BPC-157 might be used experimentally to dissect multi-organ injury mechanisms, including vascular “running” pathways that connect the gut, liver, heart, and brain.[3,16]
BPC-157, Inflammation, Angiogenesis, and Wound Healing
Wound healing and tissue regeneration represent one of the most extensively studied domains for BPC-157. In various cutaneous and soft-tissue injury models—including incisional, excisional, burn, and diabetic wounds—BPC-157 has been associated with accelerated wound closure, better granulation tissue formation, improved collagen organization, and enhanced angiogenesis.[8,17]
A comprehensive review on BPC-157 and wound healing summarized data indicating that the peptide may normalize inflammatory responses, modulate cytokine profiles, and reduce oxidative stress markers while simultaneously promoting vascular growth and extracellular matrix remodeling.[8] In fistula models (such as persistent gastrocutaneous fistulas in rats), BPC-157 treatment reportedly supported closure of otherwise non-healing tracts and restored the continuity of the affected tissues.[5]
Taken together, these findings position BPC-157 as a versatile research compound to probe the intersection of inflammation, microcirculation, angiogenesis, and matrix remodeling in complex wound and organ injury models.
BPC-157 and the Brain–Gut Axis
A growing body of work considers BPC-157 as a candidate modulator of the brain–gut and gut–brain axes. Review articles describe BPC-157 as a peptide that may “link” mucosal integrity in the GI tract with CNS function, autonomic regulation, and systemic vascular responses.[2,3,18] In this context, BPC-157 has been proposed to:
Maintain gastric and intestinal mucosal integrity under diverse stressors
Influence central neurotransmitter systems and behavioral outcomes
Modulate NO-dependent vascular tone and microcirculation in multiple organs
More recent analyses suggest that BPC-157 may help restore or stabilize bidirectional communication between the gut and brain in preclinical models, offering a unique tool to study how local mucosal damage or systemic inflammation feeds back into CNS function and vice versa.[18]
Disclaimer
The products mentioned are not intended for human or animal consumption.
Research chemicals are intended solely for laboratory experimentation and/or in-vitro testing.
Bodily introduction of any sort is strictly prohibited.
All purchases are limited to licensed researchers and/or qualified professionals.
All information provided above is for educational and informational purposes only and must not be interpreted as medical advice, treatment guidance, or endorsement of any clinical use.
References
Xue XC, Chen ZX, Zhang YM, et al. Protective effects of pentadecapeptide BPC 157 on gastric ulcer in rats. World Journal of Gastroenterology. 2004;10(4):624–628. https://www.wjgnet.com/1007-9327/full/v10/i4/624.htm PMC
Sikiric P, Seiwerth S, Rucman R, et al. Brain–gut axis and pentadecapeptide BPC 157: theoretical and practical implications. Current Neuropharmacology. 2016;14(8):857–865. https://pubmed.ncbi.nlm.nih.gov/27138887/ PMC+1
Sikiric P, et al. Stable gastric pentadecapeptide BPC 157, Robert’s stomach cytoprotection/adaptive cytoprotection/organoprotection and joint ligature/occlusion studies. Gut and Liver. 2020;14(2):153–167. https://www.gutnliver.org/journal/view.html?doi=10.5009/gnl18490 Gutn Liver
Duzel A, Vlainic J, Antunovic M, et al. Stable gastric pentadecapeptide BPC 157 in the therapy of colitis and ischemia and reperfusion. World Journal of Gastroenterology. 2017;23(48):8465–8488. https://www.wjgnet.com/1007-9327/full/v23/i48/8465.htm WJG Network
Skorjanec S, Dolovski Z, Kocman I, et al. Therapy for unhealed gastrocutaneous fistulas in rats as a model for persistent skin wounds and gastric ulcers: stable gastric pentadecapeptide BPC 157, atropine, ranitidine, and omeprazole. Digestive Diseases and Sciences. 2009;54(1):46–56. https://doi.org/10.1007/s10620-008-0332-9 Frontiers
Lojo N, et al. Effects of pentadecapeptide BPC 157 in small bowel and vascular occlusion models (intestinal ischemia–reperfusion). PLoS One. 2016;11(9):e0162590. https://doi.org/10.1371/journal.pone.0162590 Frontiers
Sikiric P, et al. Stable gastric pentadecapeptide BPC 157 in inflammatory bowel disease and ischemic colitis models. Current Pharmaceutical Design. 2011;17(16):1612–1632. https://doi.org/10.2174/138161211796196954 Frontiers
Seiwerth S, Brcic L, Vuletic LB, et al. Stable gastric pentadecapeptide BPC 157 and wound healing: angiogenesis, granulation tissue, and cytokine modulation. Frontiers in Pharmacology. 2021;12:627533. https://www.frontiersin.org/articles/10.3389/fphar.2021.627533/full Frontiers
Staresinic M, et al. Gastric pentadecapeptide BPC 157 accelerates healing of transected rat Achilles tendon and in vitro stimulates tenocyte growth. Journal of Orthopaedic Research. 2003;21(5):976–983. https://pubmed.ncbi.nlm.nih.gov/14554208/ PubMed+1
Krivic A, et al. Achilles detachment in rat and stable gastric pentadecapeptide BPC 157: promoted tendon-to-bone healing and counteraction of corticosteroid impairment. Journal of Orthopaedic Research. 2006;24(5):982–989. https://pubmed.ncbi.nlm.nih.gov/16583442/ PubMed+1
Krivic A, et al. Modulation of early functional recovery of Achilles tendon to bone healing by BPC 157 and methylprednisolone in a rat model. Inflammation Research. 2008;57(5):205–213. https://pubmed.ncbi.nlm.nih.gov/18594781/ SpringerLink+1
Tudor M, et al. Traumatic brain injury in mice and pentadecapeptide BPC 157 effect. Peptides. 2010;31(9):1650–1660. https://www.sciencedirect.com/science/article/pii/S0167011509002341 PubMed+2ScienceDirect+2
Vukojevic J, et al. Pentadecapeptide BPC 157 and the central nervous system. Neural Regeneration Research. 2022;17(3):482–489. https://journals.lww.com/nrronline/fulltext/2022/03000/pentadecapeptide_bpc_157_and_the_central_nervous.3.aspx Lippincott Journals
Jelovac N, Sikiric P, Rucman R, et al. Gastroprotective peptide BPC 157 counteracts paracetamol-induced hepatotoxicity, seizures and brain damage in rats. European Journal of Pharmacology. 1999;xx:xxx–xxx. https://www.sciencedirect.com/science/article/abs/pii/S0014299999004860 ScienceDirect
Drmic D, Kolenc D, Ilic S, et al. Celecoxib-induced gastrointestinal, liver and brain lesions in rats: counteraction by BPC 157 or L-arginine, aggravation by L-NAME. World Journal of Gastroenterology. 2017;23(29):5304–5312. https://www.wjgnet.com/1007-9327/full/v23/i29/5304.htm PubMed+2WJG Network+2
Sikiric P, et al. Stable gastric pentadecapeptide BPC 157 and NO-system relation in ischemia and reperfusion. Current Pharmaceutical Design. 2014;20(7):1126–1135. https://doi.org/10.2174/13816128113190990411 Frontiers
Tepes M, et al. Stable gastric pentadecapeptide BPC 157 therapy in multi-organ failure and major vessel occlusion models. Pharmaceuticals. 2023;16(11):1554. https://doi.org/10.3390/ph16111554 MDPI
Sikiric P, Gojkovic S, Krezic I, et al. Stable gastric pentadecapeptide BPC 157 may recover brain–gut axis and gut–brain axis function. Pharmaceuticals. 2023;16(5):676. https://doi.org/10.3390/ph16050676










 IGF DES (2mg)
IGF DES (2mg)